Principles of Isotope Fractionation
Mass-dependent isotope fractionation
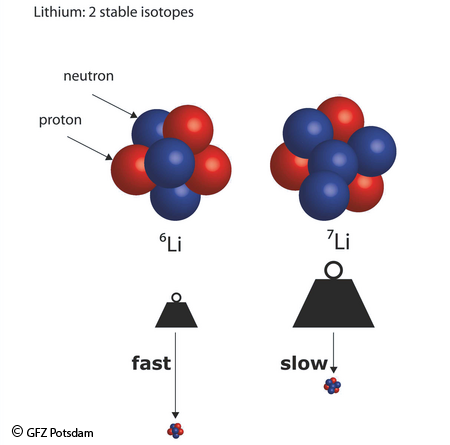
The underlying physico-chemical principle is "mass dependent isotope fractionation". The smaller the relative difference between isotopes, the larger is the relative isotope shift. This shift is induced when an element is transferred from a compartment to another which corresponds to a change in its chemical binding state. Lighter isotopes react faster (kinetic isotope effect) or enter weaker bonds (equilibrium isotope effect). The relative fractionation is largest for lithium (largest relative mass differences) and smallest for the heavy isotopes (e.g. molybdenum, uranium, smallest relative mass difference).
Isotopes in weathering

In weathering, rock is converted into soil. When soil is eroded, its products are found in solids and solutes transported by rivers. For lithium, the light isotope 6Li is partitioned preferentially into the clays that form, while the heavy 7Li moves preferentially into river dissolved Li.
Isotope fractionation through weathering processes
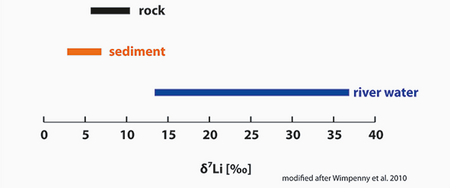
Characteristic 7Li/6Li isotope patterns are found in global rivers: light 6Li is preferentially partitioned into clays, hence its 7Li/6Li ratio is low. Heavy 7Li remains in the fluid, hence its isotope ratios 7Li/6Li are high. Shown are 7Li/6Li ratios in permil differenve to a reference material.


